  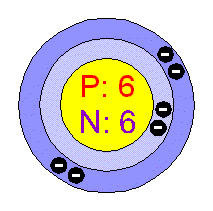
To put it another way, full outer electron shells are thought of as “happy” atoms. Make sure that the atoms are stable with the number of electrons they require. The first step is to figure out how many valence electrons there are in the atom.Ĭounting the number of valence electrons in each molecule is now complete. In these instructions, the Kelter method for drawing Lewis structures for molecules is explained. Lewis constructions can be built in a multitude of ways, so keep that in mind. Chemistry students are typically baffled by the models, however they can easily sketch Lewis structures provided the proper procedures are followed. It is also possible to utilise a Lewis structure to forecast a molecule’s geometry. Understanding Lewis structures enables you to predict the number and type of bonds that will surround an atom. In a Lewis structure, the electron density surrounding atoms is visually displayed. (It makes no difference which positions are used in which order.) Brief note on how to draw a Lewis structure: Arrangement: The dots are arranged to the right and left of the symbol, as well as above and below it, with no more than two dots on either side. The number of valence electrons in an atom is represented by the number of dots on a graph. This type of diagram (also known as an electron dot diagram, or a Lewis structure) depicts the valence electrons for a certain element in the form of a series of dots encircling the element’s symbol. Valence electrons should be depicted in a simple manner so that we can better understand how they interact with each other. Chemical bonds are produced nearly exclusively as a result of interactions between valence electrons in atoms. 
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
